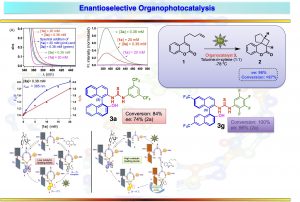
Organo-photocatalysis
Results from our group has established the use of stereoelectronic effects to manipulate excited state reactivity similar to ground state enantioselective transformations under organo-catalytic control. This opens up avenues to exploit the well-known stereoelectronic effects to control reactivity and selectivity for some of the most synthetically useful excited state transformations. Some of the recent publication from our group is listed below.
- Vallavoju, N; Sermadurai, S.; Pemberton, B. C.; Jockusch, S.; Sibi, M. P.; Sivaguru, J.* Insights into the mechanistic aspects of organo-photocatalysis mediated by thioureas. Chem., Int. Ed. 2016, 55, 5446-5451.(Selected as “important paper” and featured as a frontispiece).
- Kumarasamy, E.; Sibi, M. P.; Sivaguru, * Non-biaryl and hetero-biaryl atropisomers: Molecular templates with promise for atropselective chemical transformations”, Chem. Rev.,2015,115, 11239–11300.
- Vallavoju, N; Sermadurai, S.; Jockusch, S.; Sibi, M. P.; Sivaguru, J.* Enantioselective organo-photocatalysis mediated by atropisomeric thiourea derivatives. Angew Chem., Int. Ed., 2014, 53, 5604-5608 and Chem. 2014, 126, 5710-5714.
- Vallavoju, N. Sermadurai, S.; Jockusch, S.; Prabhakaran, M. T.; Sibi, M. P.; Sivaguru, J.* Evaluating thiourea architecture for intramolecular [2+2]-photocycloaddition of 4-alkenylcoumarins. Synt. Catal., 2014, 356, 2763-2768.